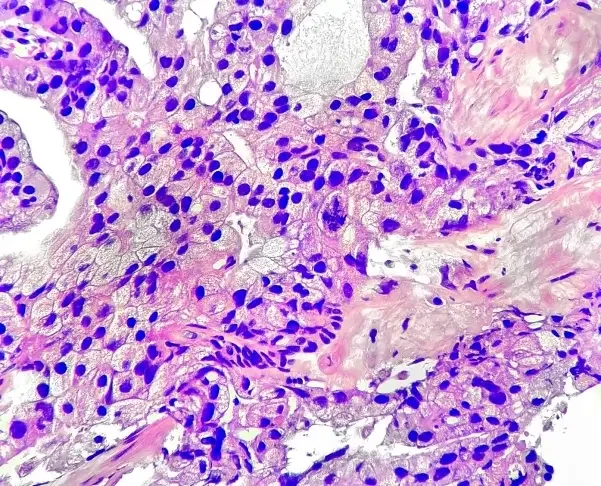
About
Artera AI is a precision medicine company leveraging multimodal artificial intelligence (MMAI) and digitized histopathology to transform how cancer therapy decisions are made. The platform's flagship product, the ArteraAI Prostate Test, is an FDA De Novo-authorized risk-stratification tool designed to guide clinicians in personalizing treatment for patients with nonmetastatic and biochemically recurrent prostate cancer. Artera's MMAI models were developed using data from NRG Oncology phase III clinical trials, incorporating diverse patient populations to reduce racial and demographic disparities in cancer treatment outcomes. The platform offers two test variants: a biopsy-based test for early-stage patients and a Post-Radical Prostatectomy (Post-RP) test for patients experiencing biochemical recurrence, helping identify those who may benefit from intensified salvage therapies such as short-term androgen deprivation therapy (ADT) combined with radiation. Designed for use by urologists, radiation oncologists, and medical oncologists, Artera AI integrates advanced deep learning with clinical data to provide actionable, evidence-backed prognostic insights. The company is also expanding its AI models into breast cancer and other oncology areas. Artera is aimed at clinical institutions and health systems seeking to bring data-driven personalization to cancer care, reducing both over- and under-treatment at scale.
Key Features
- FDA-Authorized Prostate Cancer Test: The ArteraAI Prostate Test is FDA De Novo-authorized for risk stratification of nonmetastatic prostate cancer patients to guide personalized treatment planning.
- Post-Radical Prostatectomy (Post-RP) Test: Identifies patients with biochemical recurrence following radical prostatectomy who may benefit from intensified salvage therapy, including short-term ADT combined with radiation.
- Multimodal AI (MMAI) Models: Deep learning models trained on diverse, large-scale clinical trial data (NRG Oncology phase III trials) to provide prognostic and predictive insights from digitized histopathology slides.
- Diverse & Inclusive AI Training Data: MMAI models are designed to reduce racial disparities in cancer care, with data inclusive of African American and other underrepresented patient populations.
- Expanding Oncology Coverage: Beyond prostate cancer, Artera is actively developing and validating MMAI models for breast cancer and other oncology indications.
Use Cases
- Helping radiation oncologists determine whether a prostate cancer patient should receive short-term androgen deprivation therapy (ADT) alongside radiation treatment.
- Risk-stratifying patients with biochemical recurrence after radical prostatectomy to guide salvage therapy intensity.
- Enabling clinical institutions to personalize prostate cancer treatment at scale using AI-analyzed digitized histopathology slides.
- Supporting oncology research and clinical trial design by providing AI-based prognostic biomarker data across diverse patient populations.
- Reducing over- and under-treatment of prostate cancer by providing evidence-backed predictive insights tailored to individual patient biology.
Pros
- FDA-Authorized Clinical Tool: Regulatory clearance provides clinicians and health systems confidence in the clinical validity and safety of the AI-based test results.
- Evidence-Based from Phase III Trials: Models trained on landmark NRG Oncology clinical trial data ensure the prognostic insights are grounded in rigorous, peer-reviewed evidence.
- Reduces Treatment Disparities: Inclusive AI training data actively works to close racial and demographic gaps in cancer diagnosis and treatment personalization.
Cons
- Narrow Disease Scope: Currently focused primarily on prostate cancer; broader oncology coverage (e.g., breast cancer) is still in development and validation stages.
- Enterprise/Clinical Access Only: The platform is designed for clinical institutions and health systems, with no self-service or consumer-facing tier available.
- Requires Digitized Pathology Infrastructure: Effective use depends on the availability of digitized histopathology slides, which may require upfront investment in scanning equipment or infrastructure.
Frequently Asked Questions
The ArteraAI Prostate Test is an FDA De Novo-authorized, AI-powered risk-stratification tool that analyzes digitized prostate biopsy or post-surgery pathology to help oncologists personalize treatment decisions for prostate cancer patients.
Artera uses multimodal artificial intelligence (MMAI) — deep learning models trained on digitized histopathology images and clinical data from large-scale phase III oncology trials — to generate prognostic and predictive scores for individual patients.
Artera AI is designed for clinicians including urologists, radiation oncologists, and medical oncologists, as well as clinical institutions and health systems seeking to personalize cancer therapy decisions.
Yes. The ArteraAI Prostate Test (Post-RP) was commercially launched in December 2025 and is specifically designed for patients experiencing biochemical recurrence following radical prostatectomy.
Yes. Artera's MMAI models were developed with diverse training data to account for the higher prostate cancer incidence and mortality rates in African American men, helping reduce racial disparities in treatment outcomes.