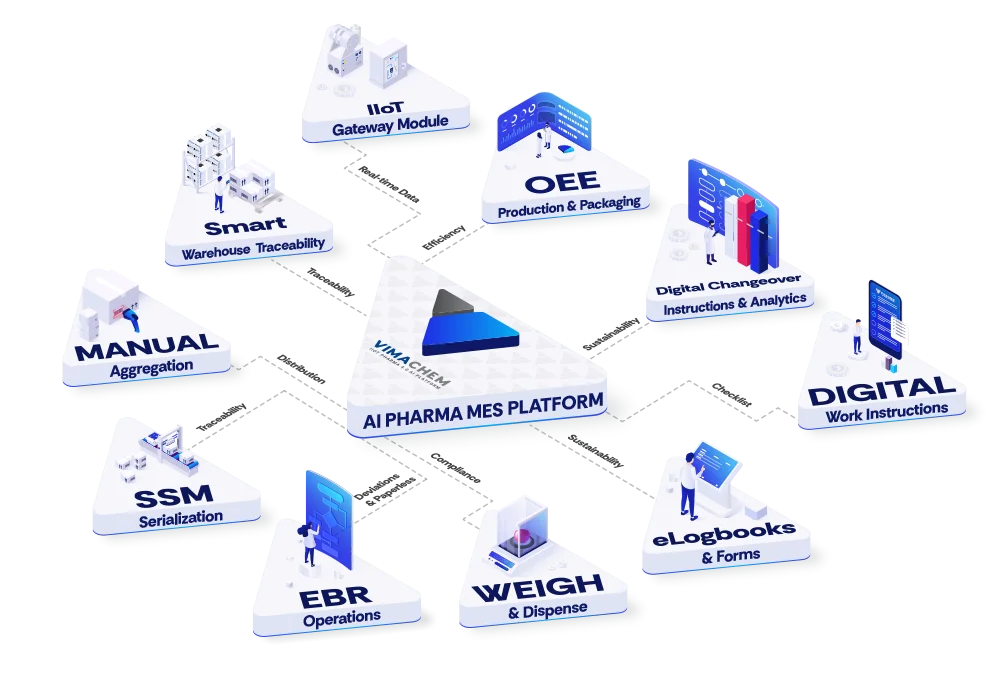
About
Vimachem is a comprehensive, AI-driven Manufacturing Execution System (MES) purpose-built for pharmaceutical and Life Sciences manufacturers. The platform embraces the Pharma 4.0 vision by combining IIoT connectivity, intelligent analytics, and GMP-compliant digital workflows into a single, configurable solution. Key modules include Electronic Batch Records (EBR) for paperless batch documentation and release, a Weigh & Dispense Module (WDM) for guided raw material handling, Digital Work Instructions for digitizing SOPs, and Electronic Logbooks for mobile-friendly compliant event logging. The Manufacturing Analytics & OEE module provides real-time equipment performance monitoring and predictive insights to minimize downtime. For track-and-trace needs, Vimachem offers a Serialization Site Manager for global packaging line serialization control, a Smart Warehouse Traceability system for managing serialized inventory across locations, and a Manual Aggregation Module. Bioprocess Monitoring Software and Digital Changeover Instructions round out the platform's capabilities. Vimachem integrates with ERP systems, SCADA, and enterprise stacks, and is backed by professional services including MES implementation, digital transformation consulting, computerized system validation (CSV), and serialization program management. It is designed for pharmaceutical manufacturers seeking to reduce compliance risk, improve audit readiness, and maximize production efficiency across their operations.
Key Features
- Electronic Batch Records (EBR): Digitize batch documentation and streamline batch release with fully paperless, GMP-compliant electronic batch records.
- Manufacturing Analytics & OEE: Monitor and optimize equipment performance in real time with AI-powered analytics and Overall Equipment Effectiveness (OEE) dashboards.
- Serialization Site Manager (SSM): Centrally control packaging line serialization across global sites to ensure regulatory track-and-trace compliance.
- Weigh & Dispense Module (WDM): Guide operators through verified raw material dispensing workflows to eliminate errors and ensure traceability at the ingredient level.
- Smart Warehouse Traceability (SWT): Manage serialized inventory across multiple warehouse locations with full visibility and audit-ready traceability.
Use Cases
- Digitizing paper-based batch records and accelerating batch release in pharmaceutical manufacturing plants.
- Monitoring and improving Overall Equipment Effectiveness (OEE) across production and packaging lines to reduce downtime.
- Managing end-to-end serialization and track-and-trace compliance for global pharmaceutical packaging operations.
- Guiding and verifying raw material weigh and dispense operations to ensure ingredient accuracy and prevent errors.
- Achieving full warehouse inventory traceability for serialized pharmaceutical products across multiple sites.
Pros
- Fully Modular Platform: Organizations can adopt only the modules they need and scale incrementally, reducing implementation risk and upfront investment.
- End-to-End GMP Compliance: Built-in support for GMP workflows, electronic logbooks, CSV, and audit trails keeps pharmaceutical operations consistently audit-ready.
- Seamless ERP & IIoT Integration: Native machine data connectivity and ERP integration bridges the gap between shop floor operations and enterprise systems.
- Dedicated Professional Services: Backed by expert services in MES implementation, digital transformation, and serialization strategy management.
Cons
- Enterprise-Level Cost: As a specialized pharmaceutical MES platform, Vimachem is positioned for enterprise clients and is likely cost-prohibitive for smaller organizations.
- Complex Implementation: Full deployment requires professional services engagement including system validation and integration work, which can extend time-to-value.
- Pharma-Specific Scope: The platform is purpose-built for Life Sciences and pharmaceutical manufacturing, making it unsuitable for other industries.
Frequently Asked Questions
Vimachem is an AI-driven, IIoT-powered Manufacturing Execution System (MES) platform designed specifically for pharmaceutical and Life Sciences manufacturers to digitize, automate, and optimize their production operations under GMP compliance.
Vimachem is built for pharmaceutical manufacturers, biotech companies, and Life Sciences organizations that need to modernize their manufacturing operations, ensure regulatory compliance, and improve production efficiency.
The platform includes Electronic Batch Records (EBR), Weigh & Dispense Module (WDM), Manufacturing Analytics & OEE, Serialization Site Manager (SSM), Smart Warehouse Traceability (SWT), Digital Work Instructions, Electronic Logbooks, Bioprocess Monitoring, Manual Aggregation Module, and Digital Changeover Instructions.
Yes, Vimachem is designed to integrate seamlessly with ERP systems, SCADA, and other enterprise software, as well as directly with shop floor machines via IIoT connectivity.
Vimachem supports GMP compliance through features like Electronic Batch Records, audit-ready electronic logbooks, computerized system validation (CSV) services, and full serialization and traceability capabilities aligned with global regulatory requirements.