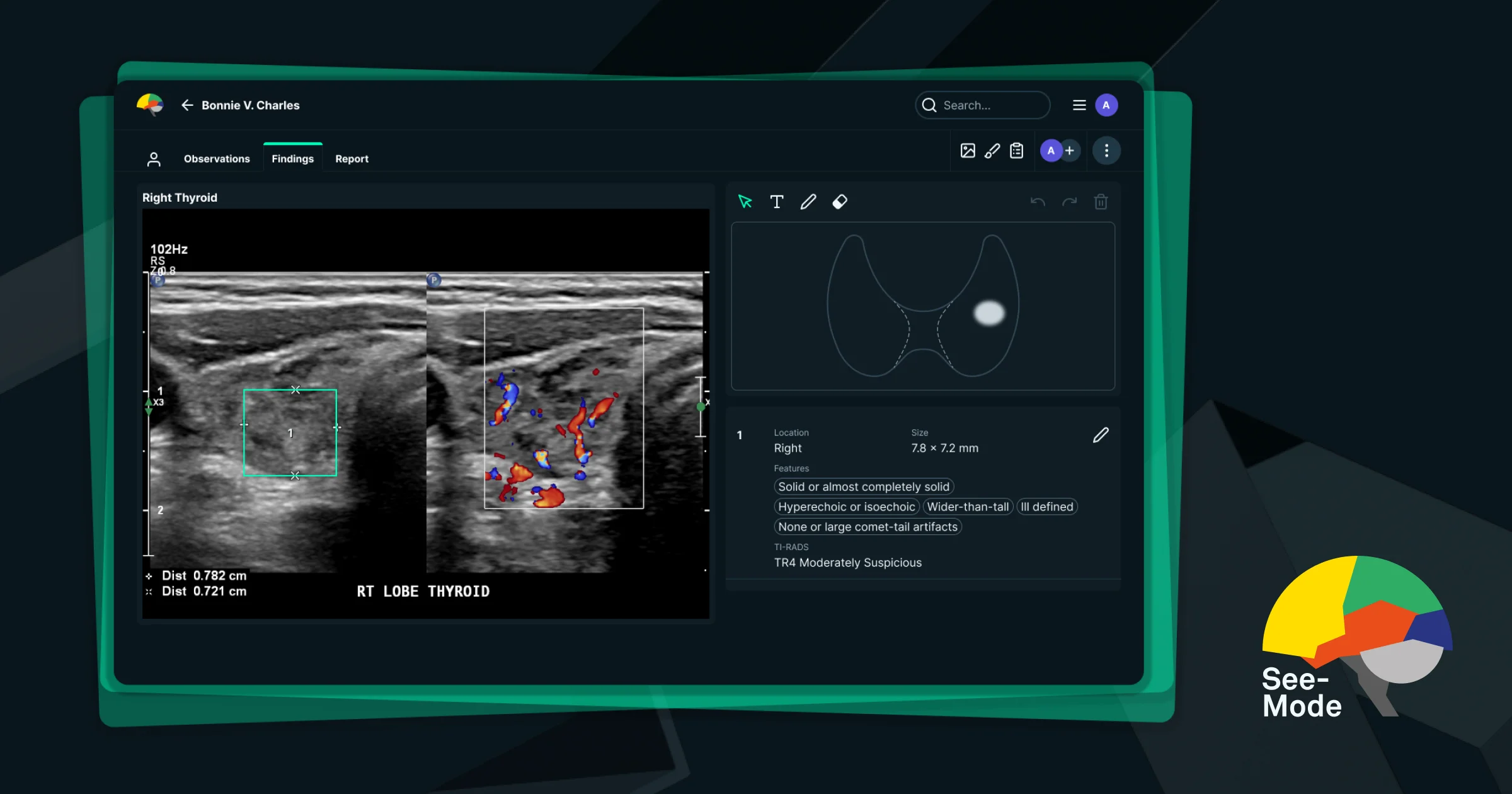
About
SeeMode Technologies is a clinical AI solution purpose-built for thyroid and breast ultrasound reporting. By automatically analyzing ultrasound images, SeeMode detects, sizes, and characterizes nodules and lesions, then produces structured radiology reports—eliminating manual steps and reducing the cognitive load on sonographers and radiologists. For thyroid imaging, SeeMode generates a complete TI-RADS sonographer worksheet and preliminary radiologist impressions, enabling faster throughput and consistent, evidence-based assessments. For breast imaging, it identifies lesions and automates BI-RADS reporting, covering both lesion characteristics and suspicion levels to reduce inter-operator variability and reporting errors. SeeMode holds FDA approval for its thyroid ultrasound AI and has demonstrated up to a 30% reduction in scan time across 12 RadNet imaging centers in early deployment. The platform is designed to integrate into existing radiology workflows, partnering with leading healthcare institutions to elevate diagnostic accuracy and consistency at scale. The tool is aimed at radiology practices, imaging centers, hospitals, and healthcare networks seeking to augment clinical staff performance, standardize reporting quality, and scale diagnostic capacity without sacrificing accuracy. SeeMode has been acquired by DeepHealth, a RadNet company, further cementing its position in enterprise medical imaging.
Key Features
- Automated Thyroid Nodule Detection & Reporting: Automatically detects, sizes, and characterizes thyroid nodules, generating complete TI-RADS worksheets and preliminary radiologist impressions.
- Automated Breast Lesion Analysis & BI-RADS Reporting: Identifies breast lesions in ultrasound images and automates full BI-RADS reporting, including lesion characteristics and suspicion level classification.
- FDA-Approved AI: Holds FDA approval for thyroid ultrasound AI, meeting regulatory standards for clinical deployment in diagnostic imaging workflows.
- Workflow Integration: Seamlessly integrates into existing radiology workflows at imaging centers and hospitals, reducing scan time by up to 30% and streamlining clinical operations.
- Consistency & Error Reduction: Reduces inter-operator variability and reporting errors, ensuring standardized, high-quality diagnostic outputs across all imaging staff.
Use Cases
- Automating thyroid nodule detection and TI-RADS classification in high-volume imaging centers to reduce sonographer workload.
- Standardizing breast ultrasound BI-RADS reporting across multi-site radiology networks to eliminate inter-operator variability.
- Generating preliminary radiologist impressions to accelerate reporting queues and reduce turnaround time.
- Supporting quality assurance in radiology departments by providing consistent, AI-verified lesion characterization.
- Scaling diagnostic capacity at enterprise healthcare organizations without proportionally increasing clinical staffing.
Pros
- FDA-Approved for Clinical Use: Regulatory approval provides confidence for enterprise and clinical adoption, distinguishing SeeMode from research-only tools.
- Proven Efficiency Gains: Demonstrated up to 30% reduction in scan time across large-scale real-world deployments with RadNet imaging centers.
- Dual Modality Coverage: Supports both thyroid (TI-RADS) and breast (BI-RADS) ultrasound reporting, offering broad value within a single AI platform.
- Institutional-Grade Partnerships: Backed by leading radiology institutions and acquired by DeepHealth/RadNet, signaling robust clinical validation and enterprise scalability.
Cons
- Limited to Ultrasound Modalities: Currently focused on thyroid and breast ultrasound only; does not cover other imaging modalities like MRI, CT, or X-ray.
- Enterprise-Oriented Pricing: Designed for institutional and enterprise radiology centers; pricing and accessibility for smaller practices or individual clinicians is unclear.
- Narrow Anatomical Scope: Coverage is limited to thyroid and breast; radiologists needing AI assistance across other anatomical regions will require additional tools.
Frequently Asked Questions
SeeMode currently supports thyroid and breast ultrasound imaging, automating TI-RADS reporting for thyroid nodules and BI-RADS reporting for breast lesions.
Yes, SeeMode's thyroid ultrasound AI has received FDA approval, making it eligible for clinical deployment in diagnostic radiology settings in the United States.
By automatically detecting and characterizing nodules/lesions and pre-populating structured radiology worksheets, SeeMode eliminates manual measurement and reporting steps. Early deployments showed up to 30% reduction in scan time.
SeeMode has been acquired by DeepHealth, a RadNet company—one of the largest outpatient radiology networks in the United States.
SeeMode is designed for radiology practices, imaging centers, hospitals, and large healthcare networks looking to augment sonographer and radiologist workflows with AI-assisted reporting.